
mRNA flu shot outperforms standard vaccine
Moderna reports its mRNA flu vaccine beat a standard shot in a late-stage trial, with stronger immune responses and a path to faster strain matching.

Moderna reports its mRNA flu vaccine beat a standard shot in a late-stage trial, with stronger immune responses and a path to faster strain matching.

Perplexity has replaced its previous Mac client with a fully native macOS app that embeds AI search into the desktop, adds system-level shortcuts, and targets power users on personal computers.
2026-05-08

Apple is preparing AirPods with built‑in cameras and on‑device AI, designed to read your surroundings, answer visual questions, and even help decide what to cook.
2026-05-08

Apple is reportedly in final testing of AirPods with infrared cameras, aimed at health and spatial tracking rather than photos or video capture.
2026-05-08

A rabid beaver attacked an 8-year-old boy fishing at Lake Henry in Mahwah, New Jersey, prompting rabies treatment for the child and public warnings from local health officials.
2026-05-08

Apple is testing camera‑equipped AirPods that pair tiny image sensors with on‑device and cloud AI, signaling a shift from audio accessory to full AI wearable platform.
2026-05-08

MV Hondius, hit by a confirmed hantavirus outbreak, is heading to the Canary Islands after dozens of passengers, including Americans, disembarked under monitoring.
2026-05-08
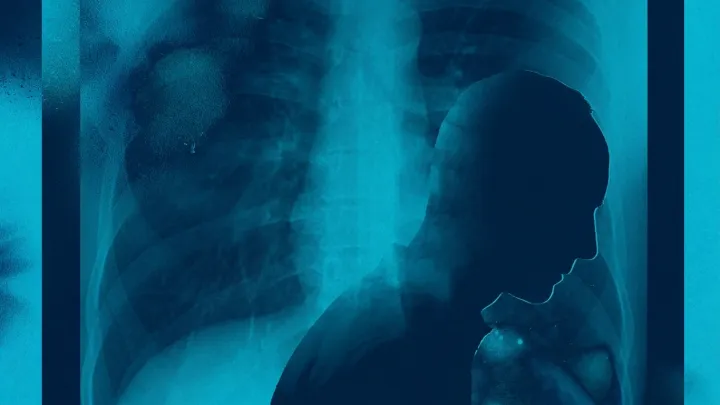
Despite targeted therapies and immunotherapy reshaping metastatic lung cancer care, about half of patients receive no treatment, blocked by late diagnosis, cost, and system bias.
2026-05-08

Perplexity has opened its Personal Computer product to all Mac users, embedding AI agents directly into the desktop to automate research, writing, and complex workflows without a browser.
2026-05-08

Dutch officials are testing a flight attendant for hantavirus after a cruise ship outbreak linked to a passenger who briefly boarded a flight from South Africa before dying.
2026-05-08

A confined cruise ship outbreak has pushed hantavirus from medical footnote to international concern as authorities in multiple countries rush to trace former passengers.
2026-05-08